Please see accompanying Full Prescribing Information and Patient Information, including BOXED WARNINGS, for additional Important Safety Information.
Important Safety Information
AMTAGVI is associated with serious and potentially life-threatening or fatal side effects, including treatment-related death, prolonged severe low blood cell counts (cytopenia), serious infections, and cardiopulmonary (heart, lung) and renal (kidney) impairment. Your healthcare provider will monitor you for these side effects during treatment.
What is the most important information that I should know about AMTAGVI?
You will likely be in a hospital prior to and after receiving AMTAGVI.
Before taking AMTAGVI, tell your healthcare provider about all of your medical conditions, including if you:
- Have any lung, heart, liver or kidney problems
- Have low blood pressure
- Have a recent or active infection or other inflammatory conditions including cytomegalovirus (CMV) infection, hepatitis B or C or human immunodeficiency virus (HIV) infection
- Are pregnant, think you may be pregnant, or plan to become pregnant
- Are breastfeeding
- Notice the symptoms of your cancer are getting worse
- Have had a vaccination in the past 28 days or plan to have one in the next few months
- Have been taking a blood thinner
Tell your doctor about all the medications you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
How will I receive AMTAGVI?
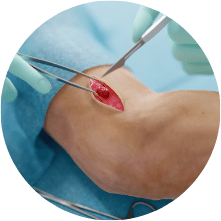
- AMTAGVI is made from your surgically removed tumor. Tumor derived T cells are grown in a manufacturing center at the end of which they number in the billions of cells.
- Your tumor tissue is sent to a manufacturing center to make AMTAGVI. It takes about a month from the time your tumor tissue is received at the manufacturing center until AMTAGVI is available to be shipped back to your healthcare provider, but the time may vary. Your AMTAGVI will be provided in 1-4 patient-specific infusion bag(s) containing 100 mL to 125 mL of viable (alive) cells per bag.
- After your AMTAGVI arrives at your treating institution, your healthcare provider will give you lymphodepleting chemotherapy to prepare your body.
- Approximately 30 to 60 minutes before you are given AMTAGVI, you may be given other medicines including:
- Medicines for an allergic reaction (anti-histamines)
- Medicines for fever (such as acetaminophen)
- Your AMTAGVI will be provided in 1 to 4 infusion bag(s) containing 100 mL to 125 mL of viable cells per bag. When your body is ready for AMTAGVI infusion, your healthcare provider will give AMTAGVI to you by intravenous infusion. This usually takes less than 90 minutes.
After getting AMTAGVI
Beginning 3 to 24 hours after AMTAGVI is given, you may be given up to 6 doses of IL-2 (aldesleukin) every 8 to 12 hours via intravenous infusion. Your doctor may discontinue IL-2 (aldesleukin) infusion any time if you have severe side effects.
You will have to stay in the hospital until you have completed the IL-2 (aldesleukin) treatment and you have recovered from any serious side effects associated with the AMTAGVI treatment.
You should plan to stay within 2 hours of the location where you received your treatment for several weeks after getting AMTAGVI. Your healthcare provider will check to see if your treatment is working and help you with any side effects that occur.
What are the possible side effects of AMTAGVI?
The most common side effects of the AMTAGVI treatment include chills, fever, low white blood cell count (may increase risk of infections), fatigue, low red blood cell count (may cause you to feel tired or weak), fast or irregular heartbeat, rash, low blood pressure, and diarrhea.
These are not all the possible side effects of the AMTAGVI treatment. Talk with your healthcare provider for more information about AMTAGVI. You can ask your healthcare provider for information about AMTAGVI that is written for healthcare professionals.
You may report side effects to Iovance at 1-833-400-46821-833-400-4682, or to the FDA, at 1-800-FDA-10881-800-FDA-1088 or at www.fda.gov/medwatch.
Please see accompanying Full Prescribing Information and Patient Information, including Boxed WARNINGS, for additional Important Safety Information.